Outsource Your iPSC Differentiation to Experienced Specialists
Leverage Trailhead Biosystems’ differentiation expertise and the power of High-Dimensional Design-of-Experiments (HD-DoE®) to convert your induced pluripotent stem cell (iPSC) lines into functional cell types for disease modeling, drug discovery and research applications.
Flexible Starting Materials
Choose the starting material that best suits your project needs:
- Partner-provided iPSC lines
- Patient-derived iPSC lines
- Gene-edited iPSC lines
- Trailhead iPSC lines
Differentiation Capabilities
We have experience with cell types across multiple lineages:
- Hematopoietic progenitor cells (HPCs)
- Endothelial cells
- Neuronal cell types including A9 dopaminergic neurons and PV+ and SST+GABAergic neurons
- Other cell type-specific differentiation upon request
How It Works
Each custom differentiation project is built around your target cell type and validation requirements. We align on starting material, define characterization criteria, and execute differentiation under controlled conditions at the scale required for your study. Final cell products are delivered with documented QC and validation data.
Trailhead Advantages
When you choose to partner with Trailhead on your custom differentiation project, you get:
- Leverage Trailhead’s HD-DoE® platform to accelerate timelines
- Deep experience with iPSC differentiation workflows across all germ layers
- Controlled differentiation with rigorous QC to support batch-to-batch consistency
- Differentiation executed at the scale appropriate for your research needs
- Scientist-to-scientist collaboration to ensure technical alignment and project success
Featured Cell Type:
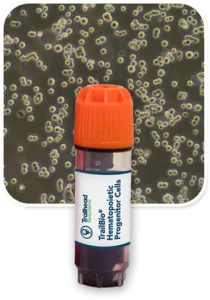
Custom Hematopoietic Progenitor Cells (HPCs)
- Our HD-DoE® platform supports controlled hematopoietic specification across control and disease model lines.
- Custom TrailBio® HPCs are suitable for differentiation into multiple blood lineages, including erythroid and myeloid pathways.
- Reach out to our team using the form below to discuss your HPC project.
TrailBio® Hematopoietic Progenitor Cells are also available for purcahse as a catalog product.
Learn more here
Ready to Start Your Project?
If you’re considering custom iPSC differentiation, tell us about your iPSC line and target cell type, and our scientific team will follow up directly to discuss your project.
For organizations that prefer marketplace procurement, Trailhead Biosystems’ custom services are also available through Scientist.com.